Content of PACT-F
Preclinical And Clinical Trials Knowledge Base on Bioavailability
In order to identify the factors which influence bioavailability, it is essential that the conditions of the trials are extensively annotated. We included all relevant information into the knowledge base, which were available in the original publications of the preclinical and clinical trials.
An extensively annotated bioavailability knowledge base is a prerequisite for the proper development of predictive models, which are able to estimate the oral bioavailability of novel compounds with high and reliable quality.
Each of the data records is composed of the chemical structure of the examined compound within that trial and up to 21 data fields which contain detailed information about the results and conditions of that trial. Each record can contain the following fields:
|
ROUTE OF ADMINISTRATION
|
SPECIES
|
|
BIOAVAILABILITY VALUES
|
STATE OF HEALTH
|
|
DOSING FREQUENCY
|
EXPERIMENTAL SOLUTION
|
|
NUMBER OF SUBJECTS
|
TYPE OF MEASUREMENT
|
|
GENDER
|
METHOD OF CALCULATION
|
|
DRUG FORMULATION
|
FEEDING CONDITION
|
|
ETHNIC GROUP
|
METABOLISM GROUP
|
|
SUBJECT STATUS
|
SUBJECT AGE
|
|
COADMINISTRATION OF ANOTHER DRUG OR COMPOUND
|
|
ROUTE OF ADMINISTRATION
The route of administration is important for a drug, so it can reach the bloodstream and will be bioavailable. Every record in PACT-F contains detailed information about how a drug was applied to a subject, such as ORAL, INTRAMUSCULAR, SUBCUTAN, RECTAL, INTRAPERITONEAL, NASAL. Since most commercially available drugs have been developed as oral medications, about 87% of all records in PACT-F describe experimental results of oral bioavailability.
|
|
SPECIES
One of the fundamental goals of PACT-F is that it identifies in nearly all records the detailed species in which the bioavailability was measured. More than 4400 records describe the results and conditions of clinical trials in humans. In more than 3600 records the detailed animal species used is specified. The majority of records is based on clinical trials in humans, followed by preclinical trials in rats, dogs, monkeys and mice.
Most of the bioavailability data in humans was measured in healthy middle-aged subjects (see also field STATE OF HEALTH). Since results from clinical trials in children, neonates or elderly humans can significantly differ from humans with average age, these groups have been further classified in PACT-F.
|
|
BIOAVAILABILITY VALUES
Within scientific publications, we have found two different descriptions of bioavailability, which were integrated into the knowledge base: qualitative and quantitative descriptions of bioavailability. The following figure shows the distribution of quantitative bioavailability values in PACT-F:
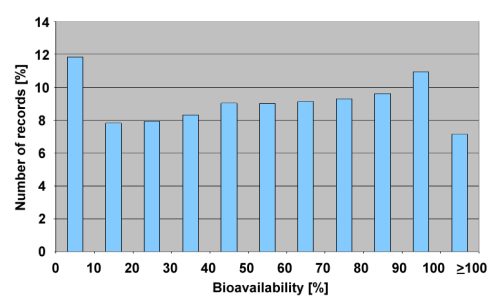
The bioavailability values were obtained from original literature or calculated from area under curve data (AUC) of the drug concentration profile. The bioavailability values in PACT-F are almost evenly distributed. The average bioavailability value was found to be 53.88%.
|
|
STATE OF HEALTH
A disease or illness can have a significant influence on the bioavailability of a drug. In some clinical trials, the bioavailability of a drug was measured in healthy subjects and ill patients. These results can be compared directly in PACT-F. Illnesses with an important effect on bioavailability can be identified and the effect of an illness can be analysed.
In order to build proper quantitative structure bioavailability relationships (QSBR) with general applicability, it is important to exclude some bioavailability results with an abnormal state of health. For example, bioavailability values from clinical trials in patients with liver dysfunction or liver disease often disagree with results in healthy patients, since normal liver function is important in bioavailability estimation.
|
|
DOSING FREQUENCY
A multiple administration of a drug can influence the bioavailability of a drug. Therefore we have collected this information whenever it was available in literature. The majority of records in PACT-F contain bioavailability values which were measured by using a single administration of a drug.
|
|
EXPERIMENTAL SOLUTION
The experimental solution in which the bioavailability of a compound was measured can have an effect on the bioavailability result.
|
|
NUMBER OF SUBJECTS
The number of individual subjects in a trial can be used to estimate the validity of an experimental bioavailability value, since there can be substantial inter-subject variability.
Furthermore this field can be used to identify trials in which oral bioavailability results of each individual subject were published in the original literature (number of subjects equals one). These data are seldom published and therefore difficult to find in the literature.
|
|
TYPE OF MEASUREMENT
In order to develop quantitative structure bioavailability relationships (QSBR) and to compare bioavailability results of different compounds properly, it is crucial to know which specific chemical entity has been measured in a trial.
In the majority of cases, bioavailability was measured based on the concentration of an unchanged drug. If a measurement is based on the concentration of an active metabolite, a contrary bioavailability value can be obtained.
For example, Perindopril is metabolized to its active metabolite Perindoprilat. The oral bioavailability based on unchanged drug yields to a value of 95%; based on the active metabolite a bioavailability of 19-20% was obtained. In other trials the concentration of unchanged and metabolised drug was measured together, leading also to a different bioavailability value of a compound.
If the detailed chemical entity was not mentioned in original literature, the applied analytical method was helpful to appraise the measured chemical entity in a trial.
Using bioavailability results without identification of the detailed chemical entity will significantly affect and reduce the quality and predictive power of QSBR-models.
|
|
GENDER
The gender of subjects can influence the bioavailability of a drug. In the majority of trials in PACT-F both genders were involved. If only one gender was used or if gender-specific differences in bioavailability were found in a trial, the gender of the subjects was included in the record.
|
|
METHOD OF CALCULATION
The bioavailability values in PACT-F were taken either directly from original publications or have been recalculated from experimental data. Most of the bioavailability values were derived from area under curve data (AUC) of the drug concentration profile.
|
|
DRUG FORMULATION
Several trials in humans or animals have analysed the influence of drug formulation on bioavailability. With the aid of this field, these differences on bioavailability can be analysed. This field can be helpful to identify drug formulations, which are favourable for a compound or a chemical structure. This information can also be used to improve the quality of a QSBR-model.
|
|
FEEDING CONDITION
The intake of food can significantly change the oral bioavailability of a drug. Several trials have analysed the influence of food on bioavailability and this field can be used to identify those trials.
|
|
ETHNIC GROUP
Some clinical trials have analysed ethnic differences in bioavailability.
|
|
METABOLISM GROUP
Some drug-metabolising enzymes like cytochrome P450 CYP2D6 exhibit a genetic polymorphism. In some clinical trials the subjects were classified into two groups like POOR METABOLISER and EXTENSIVE METABOLISER or SLOW ACETYLATOR and FAST ACETYLATOR. These types of information can be found within this field.
|
|
SUBJECT STATUS
This field describes subject-specific conditions of the examined subjects, like OBESE, PREGNANT, NON-PREGNANT, SMOKING, NON-SMOKING etc. These conditions can sometimes influence the bioavailability of a compound.
|
|
SUBJECT AGE
The age of a subject or the average age of a subject group is specified in this field.
|
|
COADMINISTRATION OF ANOTHER DRUG OR COMPOUND
The pretreatment or coadministration of a compound can largely change the bioavailability of a drug. PACT-F contains examples whereby a coadministration reduces or increases the bioavailability of a drug. The name of the coadministrated compound is stated within this field. Using this information can increase the quality and predictive power of QSBR-models.
|
|

PACT-F:
The largest
and
most
annotated knowledge base
on
bioavailability worldwide.

An
expert system
to predict human oral bioavailability
of novel drug candidates.
Recent News:
PharmaInformatic and
UNIZYME Laboratories
sign
cooperation agreement
(more)
Frequently
Asked Questions
(FAQ):
Can IMPACT-F
predict human
oral bioavailability of entirely new molecules?
Is IMPACT-F restricted to a specific
therapeutic area, such as inflammation or antivirals?
Answer
What is required to predict oral bioavailability with IMPACT-F?
At which stage of drug discovery and development should IMPACT-F be applied?
Requirements
Read all FAQs.
Expert Systems
preselect compounds which display the highest probability to become a successful drug.
Further Links:
Drug discovery
Drug development
Cheminformatic
Software
ADME
Pharmacokinetics
Computational chemistry
Molecular
modeling
Clinical trials
Preclinical
Research
Recent News:
PharmaInformatic
and
BRIDGE
BIORESEARCH
sign
cooperation agreement
(more)

PACT-F:
The largest
and
most
annotated knowledge base
on
bioavailability worldwide.

IMPACT-F
is based
on the
knowledge
of PACT-F.

PACT-F:
The largest
and
most
annotated knowledge base
on
bioavailability worldwide.

IMPACT-F
is based
on the
knowledge
of PACT-F.
PharmaInformatic provides
ADME/TOX
Knowledge Bases
and Expert Systems
to improve drug discovery
and development.





